Retatrutide: The Triple-Agonist Already Outperforming Tirzepatide in Phase 3
In this article
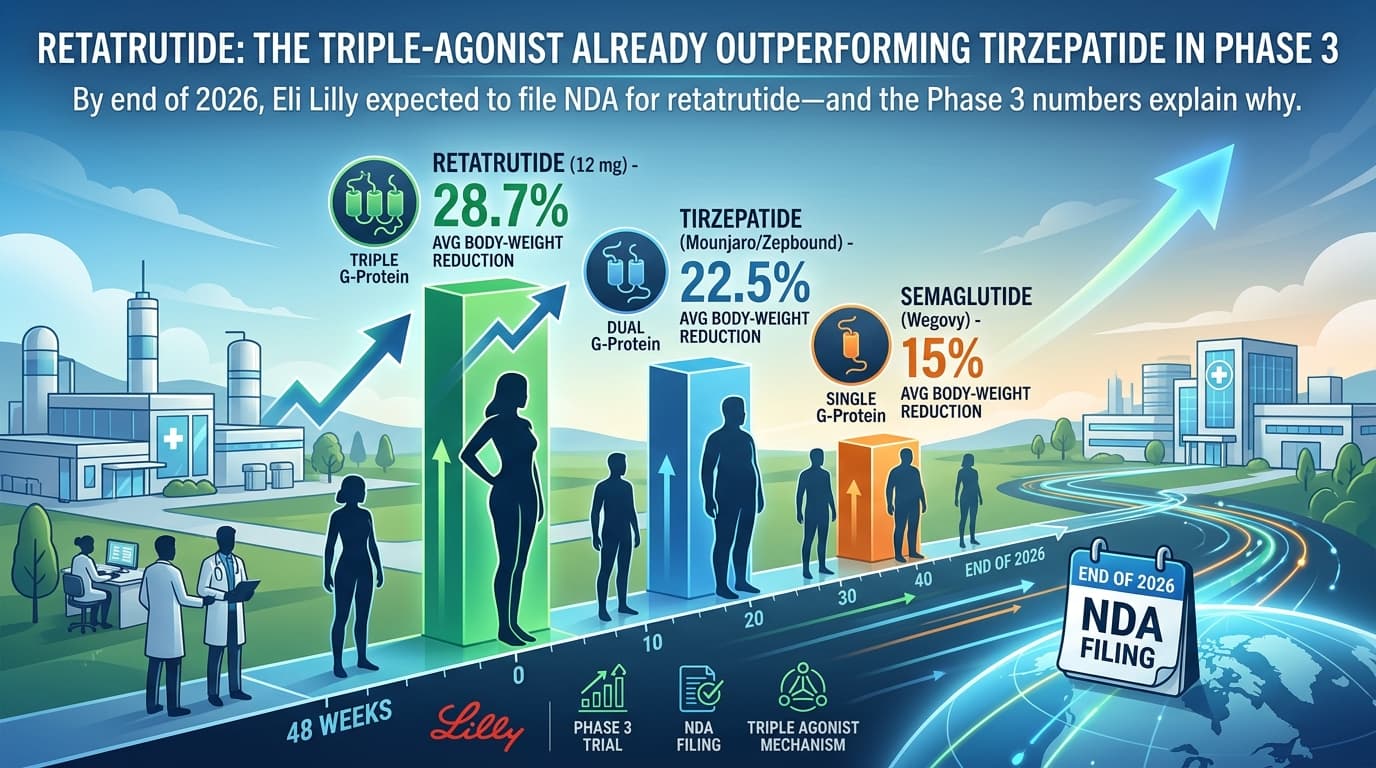
By the end of 2026, Eli Lilly is expected to file an NDA for retatrutide -- and the Phase 3 numbers explain why. In a head-to-head context, retatrutide at 12 mg has produced an average 28.7% body-weight reduction at 48 weeks, compared to tirzepatide's 22.5% (Mounjaro/Zepbound) and semaglutide's 15% (Wegovy). If those numbers hold across the rest of the trial portfolio, retatrutide will be the most effective pharmaceutical weight-loss agent ever brought to market.
For coaches, trainers, and anyone working in the obesity-and-metabolic-health space, here's what's actually happening and why it matters.
What retatrutide is
Retatrutide is an investigational once-weekly peptide that activates three receptors at once:
- GLP-1 (the same receptor semaglutide hits)
- GIP (the second receptor tirzepatide added)
- Glucagon (the third receptor retatrutide adds)
This is why the literature calls it a "triple-G agonist." The hypothesis is that hitting all three pathways simultaneously gives you appetite suppression and improved insulin sensitivity from GLP-1, gastric-emptying and energy-balance effects from GIP, and increased basal energy expenditure from controlled glucagon activation. In animal and Phase 2 data, the trio produced larger weight-loss effect sizes than any double-agonist tested to date.
Where the Phase 3 data stands as of 2026
Lilly has now reported topline results from two Phase 3 trials:
- TRANSCEND-T2D-1 (March 2026, type 2 diabetes): Retatrutide met the primary and all key secondary endpoints. A1C reductions up to 2.0%. Weight loss up to 36.6 lbs (16.8%) at 40 weeks vs placebo.
- TRIUMPH-4 (December 2025, obesity + knee osteoarthritis): Up to 71.2 lbs average weight loss, plus substantial relief from osteoarthritis pain in adults with obesity or overweight without diabetes.
Seven additional Phase 3 trials are scheduled to complete in 2026, covering obesity, type 2 diabetes, obstructive sleep apnea, chronic low back pain, cardiovascular and renal outcomes, and metabolic dysfunction-associated steatotic liver disease (MASLD). NDA filing is targeted for Q4 2026.
Why this matters for fitness professionals
GLP-1 receptor agonists have already changed the landscape for trainers, nutrition coaches, and anyone working with overweight or obese clients. Retatrutide will accelerate that shift, not reverse it. Three things to think about:
- The lean-mass question gets sharper. With weight-loss effect sizes pushing into the 25-30% range, lean-mass preservation becomes the dominant performance variable. Resistance training and adequate protein during the active loss phase aren't optional add-ons -- they are the difference between losing 30% body weight as fat plus 5% as muscle, vs losing 30% as fat with 1-2% lean loss. Coaches who build that protocol now will own this market.
- Plateau and discontinuation coaching becomes a real specialty. Across the GLP-1 class, weight regain after discontinuation is well-documented. Retatrutide will be no different -- the question is what protocol prevents the rebound. This is a coaching gap, not a pharmaceutical gap.
- The pain-and-mobility angle is new. TRIUMPH-4 was specifically designed around osteoarthritis. If retatrutide gets a co-indication for joint pain, the trainer's value proposition changes -- you're not just helping a client lose weight, you're helping them get to the gym with less pain.
Where retatrutide isn't yet
It is investigational. As of April 2026 it is not FDA-approved. It is not commercially available through legitimate prescription channels. Compounded versions exist in the gray market and almost universally sit outside Lilly's quality control.
The trial program is robust enough that approval looks likely, but "likely" is not "approved." Don't recommend, source, or coach around retatrutide as if it's already on the shelf -- it's not, yet.
The bottom line
Retatrutide is on track to redefine pharmaceutical weight loss within 18 months. The fitness industry has roughly that long to figure out the protocols that go alongside it: lean-mass preservation, post-discontinuation coaching, and pain-and-mobility integration. Get those right and the GLP-1 / triple-G era is a tailwind, not a threat.
Written with research from Lilly investor releases, PharmExec, ClinicalTrialsArena, and PubMed.
EvoFit Team
AI-powered fitness science, nutrition research, and coaching strategies for the modern fitness professional.


